Transcatheter aortic valve replacement (TAVR) is a minimally invasive procedure used to replace the aortic valve in people with severe aortic stenosis. In the past, valve replacement required open heart surgery. With TAVR, our doctors use a catheter (thin tube) to fit a new valve inside the diseased valve.Because surgery is not needed, patients recover faster and spend less time in the hospital. Most patients notice dramatic improvement within a few days or weeks.
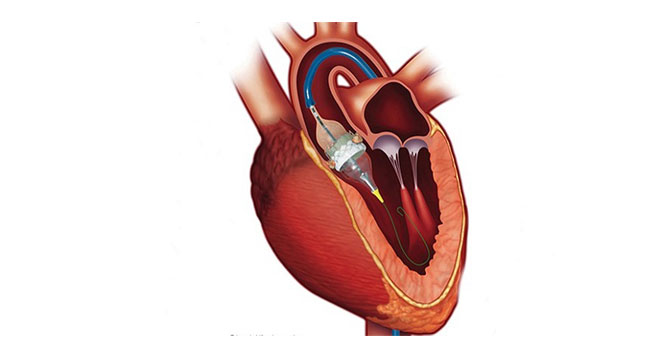
TAVI/TAVR isperformed through groin, axilla, or neck vessels under sedation or a short duration ofanesthesia. Pre-procedural CT scan and detailed echocardiography studies help in offering tailor-madesolutions for each procedure.Our doctors insert a catheter into an artery using a needle puncture. The 1-2 hourprocedure is guided by fluoroscopyand cine recording in thecatheterization laboratory. There are two main approaches:Transfemoral (through the femoral artery in the groin) and Transapical (through an artery in the chest or the tip of the heart’s left ventricle (apex)). Once the catheter has been inserted and reaches the aortic valve, the doctor places the collapsible replacement valve. As it expands, it pushes the old valve’s leaflets out of the way. Before removing the catheter, our team evaluates how the new TAVR valve functions and ensures it is in the correct position. If needed, the valve can be repositioned.
TAVR is for patients with severe aortic stenosis. Stenosis is a narrowing of the valve opening caused by calcium buildup or scarring of the valve’s leaflets.The narrow valve opening restricts blood flow from the heart to the aorta. This requires the heart to work harder and can cause symptoms such as shortness of breath, chest pain and heart failure. For these patients, TAVR can greatly improve their quality of life.Originally, TAVR was only approved for patients who could not tolerate surgical aortic valve replacement. In 2019, TAVR was approved by the Food and Drug Administration (FDA) for all patients. This approval was based on clinical trial results showing TAVR outcomes were equal to or better than open surgical valve replacement.TAVI obviates the need for Open Heart Surgery or “Sternotomy” and the patients are discharged from the hospital within 72 hours of the procedure.
Research suggests the valve may last up to 10 years.If the valve were to fail, our doctors can replace it. This can be done surgically or with a new TAVR valve inserted inside the old one.
In the last 20 years, TAVI has become immensely popular with patients and cardiologists alike,TAVI has replaced surgical replacementof heart valves as the preferred mode of treatment. By 2025, it is expected that 280000 TAVI procedures will be performed annually.
Read More: One Lung Anaesthesia/Single lung ventilation